
Concrete is a mixture of aggregates (sand and rocks), water and a material that binds them all together — cement. Over time, the water and cement chemically bonds together through a process called hydration, hardening and holding all the sand and rock together. This means that concrete behaves like a paste when water is first mixed into the cement - malleable into any shape. The ability to take on any shape before hardening is what makes concrete such a great material. (Portland Cement Association, 2015)
http://www.cement.org/images/default-source/c-cbasics/components.jpg?sfvrsn=2
This image shows the typical composition of concrete.
http://www.cement.org/images/default-source/c-cbasics/components.jpg?sfvrsn=2
This image shows the typical composition of concrete.
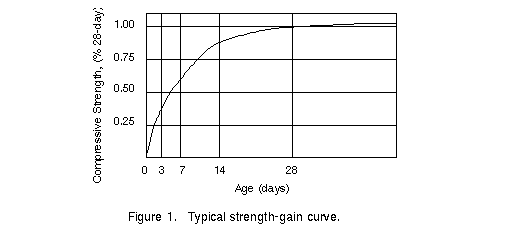
http://www.ce.memphis.edu/1101/notes/concrete/graphics/conc3.gif
This graph shows the strength of concrete over time.
Cement is the main ingredient in concrete. Concrete is mainly made of limestone, clay and a combination of other minerals. These materials are collected and are then crushed into small pieces of about 6cm in width. Then, they are mixed with other materials and put into a kiln where temperatures reach 1450º Celsius to form new compounds. Finally, these compounds are crushed into a fine powder which is cement. (Portland Cement Association, 2015)
The main components in the final product of Portland cement is a mixture of silicates and oxides:
Belite (2CaO·SiO2) Alite (3CaO·SiO2)
Tricalcium aluminate (3CaO·Al2O3) Brownmillerite (4CaO·Al2O3·Fe2O3)
All the molecules in cement chemically react with water to form new compounds. The hydration of portland cement is a exothermic reaction. The heat generated from the hydration of cement increases the temperature of the concrete, which means that the reaction is exothermic. I will focus on the main product of the hydration process of Portland cement, calcium silicate hydrate. This compound is primarily responsible for the strength in concrete. Since energy is released from this reaction, it means that the reaction is exothermic. (Portland Cement Association, 1997)
Tricalcium Silicate + Water → Calcium Silicate Hydrate + Calcium Hydroxide + heat
2 Ca3SiO5 + 7 H2O → 3 CaO · 2 SiO2 · 4 H2O + 3 Ca(OH)2 ∆H=173.6 kJ
The main components in the final product of Portland cement is a mixture of silicates and oxides:
Belite (2CaO·SiO2) Alite (3CaO·SiO2)
Tricalcium aluminate (3CaO·Al2O3) Brownmillerite (4CaO·Al2O3·Fe2O3)
All the molecules in cement chemically react with water to form new compounds. The hydration of portland cement is a exothermic reaction. The heat generated from the hydration of cement increases the temperature of the concrete, which means that the reaction is exothermic. I will focus on the main product of the hydration process of Portland cement, calcium silicate hydrate. This compound is primarily responsible for the strength in concrete. Since energy is released from this reaction, it means that the reaction is exothermic. (Portland Cement Association, 1997)
Tricalcium Silicate + Water → Calcium Silicate Hydrate + Calcium Hydroxide + heat
2 Ca3SiO5 + 7 H2O → 3 CaO · 2 SiO2 · 4 H2O + 3 Ca(OH)2 ∆H=173.6 kJ